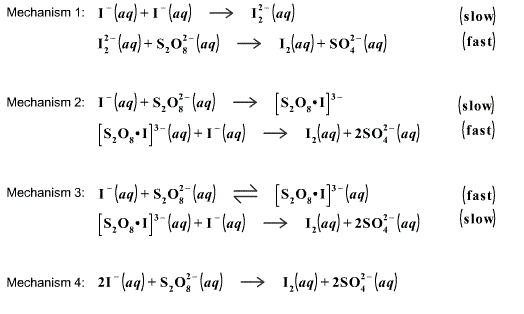SOLVED: proposed mechanism for the iodine clock reaction is shown below: Iodine Clock Reaction Mechanism IO3 + HSO3 S0 + HIO2 k =2.95 x 10-'M-Is-1 HIO2 + I- + H+ + 2HOI

SOLVED: Reaction Rates Concentrations Effect of Reactant Concentrations Demo: "lodine Clock" Reaction lodateVariation I03 + 3 HSO3 > 1-+ 3 S042-+3 Ht (slow) I03 +51 + 6 Ht> 3 12 + 3

Iodine Clock Reaction We will begin by describing a proposed reaction mechanism for the iodine clock reaction. There are several variations to this reaction, - ppt download
What is the reaction order with respect to [S2O8 2-] and [I-] in a persulfate-iodide reaction? - Quora
![SOLVED: The rate law for the iodine clock reaction is given by: Rate = k[IO3- ] [I- ] 2[H+]^2a)This reaction is third order with respect to H+.b)This reaction is first order with SOLVED: The rate law for the iodine clock reaction is given by: Rate = k[IO3- ] [I- ] 2[H+]^2a)This reaction is third order with respect to H+.b)This reaction is first order with](https://cdn.numerade.com/ask_previews/494c958b-2794-4a7f-861f-022f5f445939_large.jpg)


![How to do lab report [Exp 004] Rates of Reaction for Iodine Clock Reaction - YouTube How to do lab report [Exp 004] Rates of Reaction for Iodine Clock Reaction - YouTube](https://i.ytimg.com/vi/L1CtBY_xmZs/maxresdefault.jpg)